Ca atomic radius5/28/2023  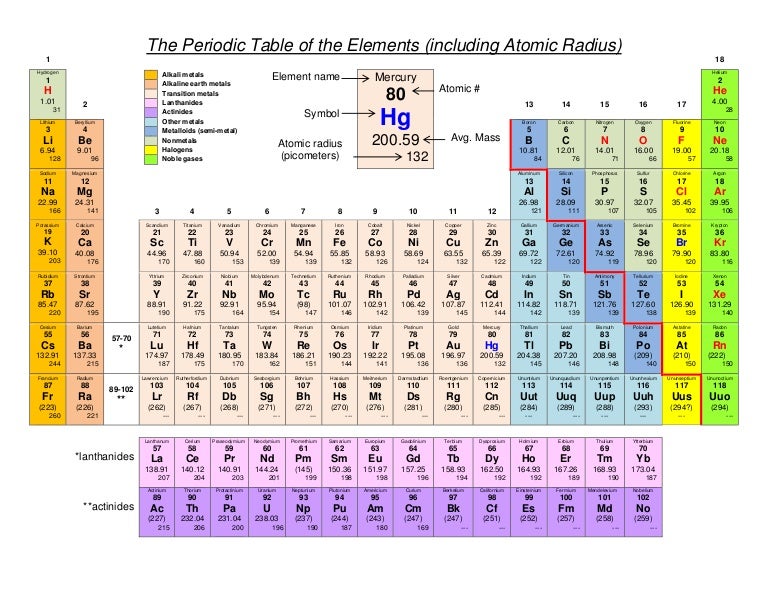
Which is not as far from the result of the orthodox method of obtaining the distance of the edge. Anyways, orking back from my original problem. I saw that I made a mistake in my original determination of one of the sides of the cube. Wait, turns out I actually wasn't my way was just a longer way of executing the steps to achieve the desired volume. Now I'm wondering why my original result, with 2r + x, was wrong. Up to date, curated data provided by Mathematica 's ElementData function from Wolfram Research, Inc. But I think I saw something similar to the result I've obtained in my book. I never thought of making d its own variable I just thought to make d = 2r + x. This image shows the geometry of the crystal: Your determination of the edge length, d, of the crystal is suspect. The covalent radius of a chlorine atom, for. 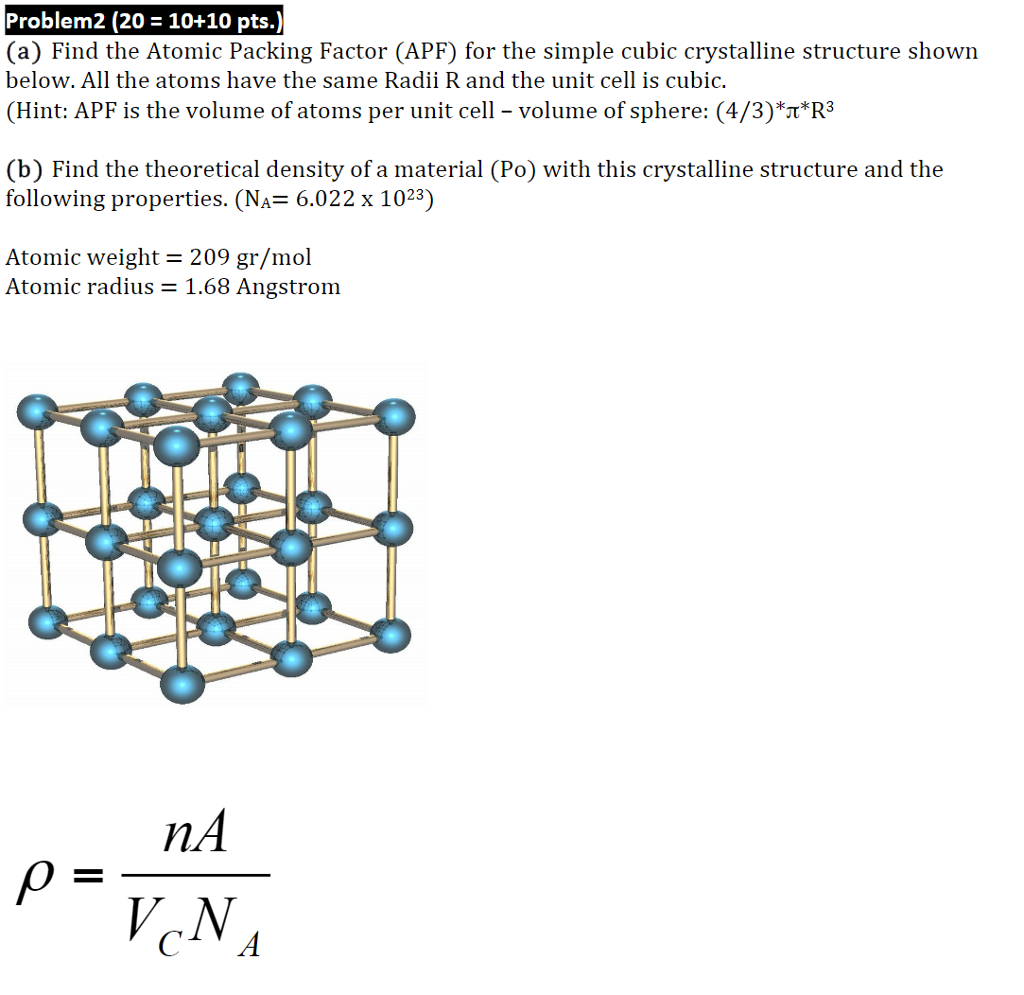
Can someone tell me what I'm doing wrong with my calculations? The size of an atom can be estimated by measuring the distance between adjacent atoms in a covalent compound. I checked, and the density of solid calcium is 1.54 g/cm 3. On moving from top to bottom in a group, the atomic radius increases. Now I know this isn't the correct answer. This is because sodium and potassium are elements of group 1. Next, I did the volume and mass calculations, the errors of which I am sure are the result of my distance calculations. This is how my distance calculations turned out, and I'm pretty sure I screwed something up. I would use the Pythagorean theorem to equate the squares of those two distances to 4r, which is the longest distance inside a cube. Then I would have to figure out the distance between two corners diagonally opposite each other on any given face of a cube, given by the square root of 2(2r +x) 2. I started out with the edge of the cube, which I presumed to be 2r + x (some unknown distance I needed to figure out). The longest distance inside the cell from one end to another is 4r. Okay, I am sure I messed up at part (c), where I'm calculating the length of the unit cell's edges in relation to the atomic radius. (a) How many Ca atoms are contained in each unit cell? (b) How many nearest neighbors does each Ca atom possess? (c) Estimate the length of the unit cell edge, a, from the atomic radius of calcium (1.97A). "Calcium crystallizes in a body-centered cubic structure. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
